News Details
Organismal shape can be decomposed into different components that carry distinct evolutionary signals. Functionally relevant shape components often are under natural selection, whereas “constructional details,” such as the contributions of the different bones to overall shape, tend to evolve more neutrally and thus better trace phylogenetic history.
A major aim of evolutionary biology is to reconstruct the phylogenetic relationships among taxa, living and extinct. Knowing how taxa on specific branches of the Tree of Life are related is necessary to test hypotheses about adaptation and developmental constraints, about evolutionary novelties, and evolutionary stasis or lability of traits, to name a few. By and large, scientists have mapped species onto this tree fairly well, but the phylogenetic positions of many fossil taxa remain unresolved – and even those of some living groups remain controversial (e.g., turtles). In the absence of informative genetic data, highly derived traits and morphological similarities owing to ecological similarities (so-called homoplasies) complicate efforts to reconstruct their phylogenetic relationships. Functionally relevant aspects of organismal morphology are often subject to natural selection and, hence, do not reliably trace phylogenetic history the way that neutral genetic markers do. For example, based on genetic data we now know that the African papionin monkeys – which include mangabeys, baboons, and relatives – are related in a way quite different from what morphological studies had long suggested. This incongruence also conceals the phylogenetic position of closely related fossil papionins for which no genetic data exist.
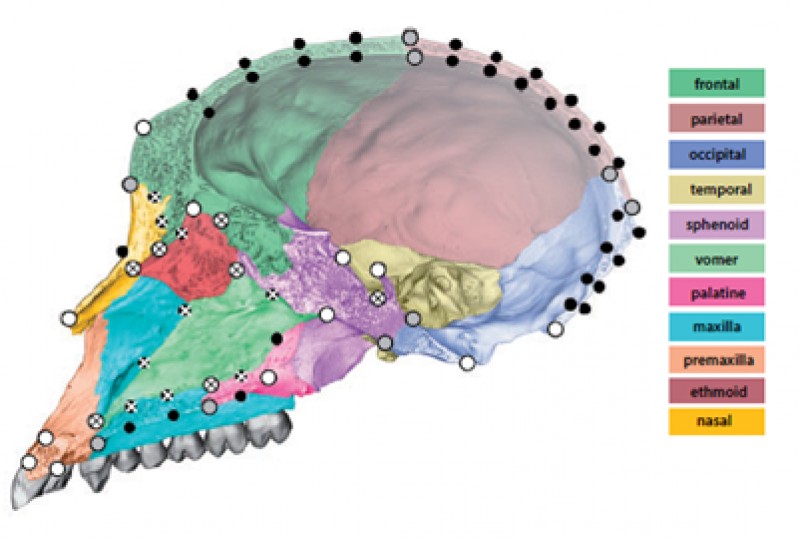
To solve this problem, Mitteroecker, Grunstra and colleagues devised a set of new geometric morphometric and statistical methods to isolate “neutral traits” from functional traits in morphological data. Such neutral phenotypic traits could then be used to reconstruct phylogenetic relationships. The researchers focused on the cranium for its morphological complexity and because cranial fragments feature prominently in the fossil record. Furthermore, the vertebrate cranium accommodates many functionally relevant sensory organs, it protects the brain, and it houses the feeding apparatus. As such, the cranium is well known to reflect a mosaic of adaptations. However, it is also composed of multiple bones that together make up a functional whole, with no individual cranial bone performing any of the aforementioned functions on its own. The relative extent to which individual cranial bones contribute to their larger structure may thus be largely irrelevant; important is the shape on a larger scale that they contribute to (see figure 1 below). Overall, large-scale shape and the “constructional details”, or small-scale shape features, may thus carry different evolutionary signals, the researchers hypothesized.

Figure 1. Schematic drawing of primate crania in midsagittal view. The overall shape of the cranium is highly similar within each of the two species, but the relative dimensions of the individual cranial bones (some of which are displayed in color) vary considerably. Many functions performed by the cranium, such as sensing or masticating food, involve multiple cranial bones simultaneously and so overall size and shape are likely functionally important and subject to natural selection. However, the relative contributions of individual bones to these morphologies is functionally much less relevant and may evolve more neutrally.
The authors tested these predictions in a sample of papionin monkeys (which also include macaques) that are well-known for their cranial homoplasies. In other words, these monkeys’ cranial morphology obscures their underlying phylogenetic relationships. Consistent with their hypotheses, Grunstra and colleagues found that overall cranial shape, ignoring individual bone contributions (see figure 2 below), and other large-scale shape features had a weak association with the monkeys’ molecular phylogeny, while being more strongly correlated to various ecological and dietary variables. By contrast, the relative contributions of individual bones performed surprisingly well in reconstructing phylogenetic relationships and had a weaker association with ecology and diet.

Figure 2. The decomposition of total midsagittal cranial shape into an “outline shape” component that captures overall shape and ignores individual bone contributions, and a “residual shape” component that is standardized for outline shape variation and therefore only captures the relative contributions of the individual bones.
The ability to decompose organismal shape into components that correspond to more functional and more neutral traits, respectively, improves on traditional approaches to reconstruct phylogenetic history from morphology. In paleontology, where phylogenetic reconstruction is of major importance but where organic remains for molecular phylogenetic analysis are typically not available, these new methods may prove fruitful to decipher relationships among fossil taxa. Furthermore, this novel shape decomposition opens up exciting new possibilities for testing evolutionary hypotheses about a range of ontogenetic and evolutionary processes.
Author: Nicole Grunstra
Publication:
Nicole D S Grunstra, Silvester J Bartsch, Anne Le Maître, Philipp Mitteroecker, Detecting Phylogenetic Signal and Adaptation in Papionin Cranial Shape by Decomposing Variation at Different Spatial Scales, Systematic Biology, 2021 ; syaa093
https://doi.org/10.1093/sysbio/syaa093

